A B S T R A C T
The aim of these studies was to determine the reduction in pasture infectivity likely to be achieved by the supplementation of grazing sheep with BioWorma®,
a product containing the chlamydospores of the nematophagous fungus Duddingtonia flagrans strain IAH 1297. Four placebo-controlled trials were conducted
between 2009 and 2013 in sheep in different climatic regions of New South Wales and Queensland, Australia and across several seasons. The effectiveness
of BioWorma was assessed by total worm counts in tracer sheep placed in paddocks grazed by parasitised sheep which were fed a daily supplement with
and without BioWorma under group-feeding conditions. Further proof of concept was obtained by assessing the worm burdens and weight gains of the parasitised
sheep, as well as the number of anthelmintic (“salvage”) treatments required when faecal egg counts exceeded a threshold level.
Keywords: BioWorma, Duddingtonia, Nematophagous fungi, Parasitic nematodes, Nematode control
Authors: Kevin Healey, Chris Lawlor, Malcom R. Knox, Michael Chambers, Jane Lamb
Gastrointestinal nematode parasites (GIN) are of great concern for producers of sheep and other grazing livestock worldwide. Several species of nematodes
affect sheep including Haemonchus spp., Teladorsagia (Ostertagia) spp., and Trichostrongylus spp. although differences in prevalence and abundance
occur in different geographic locations due to local ecological and climatic zones. Infection with GIN results in significant losses in productivity
and reproductive performance and impacts negatively on animal health, causing diarrhoea, anaemia and, in some cases, death. In higher rainfall areas
of Australia where sheep contribute to on-farm profitability through wool and meat production, GIN parasites severely impact production if effective
control measures are not undertaken. In Australia, internal parasites were identified as the highest cost disease of sheep (Sackett et al., 2006) and
the annual cost associated with parasitic diseases in sheep and cattle has been estimated at A$1 billion (Roeber et al., 2013).
Since the 1960s, the regular appearance and availability of a number of effective anthelmintic chemicals has provided a ready solution to this problem. Over the past 25 years however, it has become apparent that the regular application of anthelmintic chemicals has led to the development of strains of the major pathogenic nematode species that are resistant to all of the currently available anthelmintics (Besier and Love, 2003; Kaplan and Vidyashankar, 2012; Playford et al., 2014, Lamb et al., 2017). It is also evident that the rate of development and registration of “new” anthelmintics is not keeping pace with the rate of emergence of strains of nematodes resistant to available anthelmintics (Hennessy, 2000). Concerns over the increasing prevalence of anthelmintic resistance and the increasing consumer demand for products with minimal chemical inputs has led researchers to place greater emphasis on finding non-anthelmintic means of combating this problem (Gill and Le Jambre, 1996; Knox et al., 2012).
Unlike nearly all other methods for parasite control in livestock, which are aimed at the parasitic stage within the host animal, biological control methods can be targeted at the free living parasitic stages on pasture (Knox, 2003; Waller, 2006). Nematode trapping fungi are found in both natural and agricultural soils (Duddington, 1951; Fernandez et al., 1999) where they live saprotrophically or predatorily (in the presence of nematodes) (Bogus et al., 2005). A large number of nematode-destroying fungi have been identified to date however only a few have been studied for use in controlling parasitic nematodes in animals (Waller and Larsen, 1996; Knox, 2003). Duddingtonia flagrans is currently the most studied fungus due to the ability of its thick walled chlamydospores to survive gut passage, germinate and grow rapidly in fresh faeces and its avid nematophagous capacity (Larsen, 1999; Knox, 2003).
A number of studies using D. flagrans have reported success in its use to control GIN (Waller et al., 2001; Fontenot et al., 2003; Chandrawathani et al., 2004; Paraud et al., 2005). The chlamydospores of D. flagrans can be added to animal feed (Mendoza de Gives et al., 2006) and pelleted feed (Hernandez et al., 2016) and pass through the animal’s gastrointestinal tract, which ultimately leads to decreased numbers of pre-parasitic nematode larvae in faeces and on the surrounding pasture (Ojeda-Robertos et al., 2009; Paz-Silva et al., 2011). When used in combination with other control strategies, predacious fungi have the potential to decrease the reliance of farmers on anthelmintics.
Several studies have shown the passage of D. flagrans chlamydospores into the faeces of sheep after oral drenching (Larsen, 2000) and efficacy has been demonstrated in a number of field trials where D. flagrans spores were fed to sheep (Githigia et al., 1997; Knox and Faedo, 2001; Peart, 2002; Fontenot et al., 2003; Chandrawathani et al., 2004; Gomez-Rincon et al., 2006; Santurio et al., 2011). In this report, a series of field trials were carried out to investigate the effect of supplementation of BioWorma, a product containing the chlamydospores of an Australian isolate of the fungus D. flagrans, on GIN burdens in sheep in different regions and seasons in Australia.
In these placebo-controlled trials the following products were used:
In each of the 4 trials, the groups of sheep used had one of two designated roles:
In each trial a pair of matched paddocks (Paddock 1 and 2) was used to graze sheep. Paddock 1 was grazed with a group of seeder sheep which received a daily supplement of the placebo (Control Group) whilst Paddock 2 was grazed by a matching group of seeder sheep that received an equivalent amount of a daily supplement of BioWorma (BioWorma Group). Individual faecal egg counts (FECs) were conducted regularly (weekly or fortnightly) to monitor and confirm patent infections, with samples collected per rectum. FECs were conducted according to a modified McMaster method (Hutchinson, 2009) with sensitivity of 40 eggs per gram (2.5 g samples examined). Individual bodyweights were monitored monthly using electronic livestock scales (verified before and after weighing using calibrated test weights). The daily supplements were prepared using verified electronic scales (to 0.01 kg) and administered in a group setting in covered troughs at the same time each day. Any uneaten supplement was removed daily and weighed.
Trial paddocks chosen for the 4 independent studies were located in well-known sheep grazing regions of Australia and had not been grazed by sheep or goats for a minimum period of 2 months prior to introduction of seeder sheep/commencement of the study. Trials were conducted over a range of climatic conditions and the pastures were typical of those used to graze sheep in the region, with stocking density equivalent for each paddock. In Trial 1 the stocking density was higher than the regional average to increase the likelihood of larval ingestion (approx. 20 sheep per 0.5 ha) whilst in Trials 2–4, stocking densities conformed to regional averages. Throughout all trials there was sufficient pasture available to maintain liveweight gain in sheep at the stocking density used without need for supplementation, according to the established regional requirements. In Trial 1, paddocks were confirmed free of contamination by pre-trial grazing with worm-free tracer animals (group geometric mean FECs of pre-trial tracers were 5.4 and 0.1 epg for paddocks 1 and 2 respectively). In all subsequent trials paddocks had a similar history of grazing and period of rest (no grazing) to those in Trial 1 and hence pre-trial tracers were considered unnecessary.
After the seeder sheep had grazed the paddocks for two months, the degree of pasture contamination by infective nematode larvae was assessed by grazing paddocks with worm-susceptible tracers for a period of 3 weeks. Prior to grazing, the tracers were confirmed to be free of nematode infections by treatment with a broad-spectrum, short-acting non-residual anthelmintic combination drench and subsequent individual FEC. Anthelmintics were administered orally and based on label recommendations and individual bodyweight. Tracers were maintained in pasture-free pens for a period of 2 weeks to allow dissipation of anthelmintics prior to relocating tracers onto the trial paddocks. Tracers were subsequently allocated randomly to two groups (Group A and B). Group A grazed on the Control paddock (Paddock 1) whilst Group B grazed the BioWorma paddock (Paddock 2). The quantity of supplements provided (placebo and BioWorma) were proportionally increased while the tracers were grazing. The tracer animals were then removed from paddocks to raised pens to allow any worm burdens to mature. The level of infection in tracer sheep was determined by conducting total worm counts (TWCs) after sacrifice, on aliquots collected from gut washings. In Trials 2–4 the seeder sheep continued to graze the trial paddocks for a further two months at which time another group of tracers were introduced as above and their TWC was determined. The additional groups of tracers were designated groups C and D (grazing Control and BioWorma paddocks respectively).
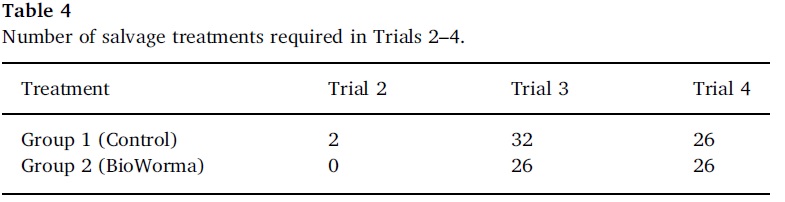
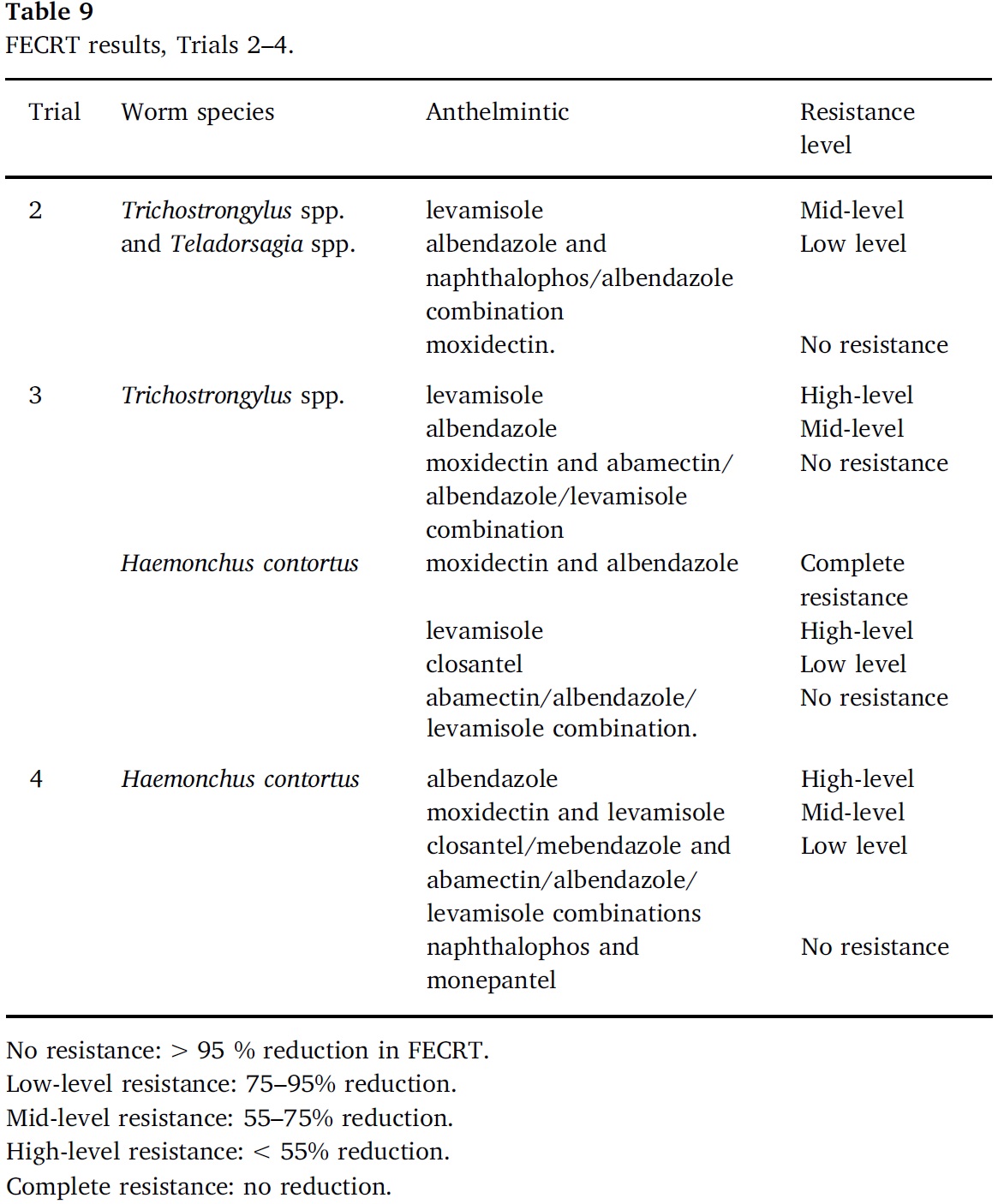
A salvage treatment was administered to any seeder sheep throughout the study if FECs (monitored weekly/fortnightly) exceeded a trigger threshold and/or veterinary indications based on ethical considerations, to prevent occurrence of clinical parasitosis between visits to trial sites. Salvage treatments consisted of a short-acting non-residual anthelmintic administered orally based on label recommendations and individual bodyweight. In Trials 3 and 4 the FEC trigger threshold was reduced because Haemonchus spp. was predominant amongst the species present and the more remote trial locations meant that veterinary oversight would be less frequent. The number of salvage treatments required throughout each trial formed an integral assessment of treatment efficacy.
The trials were conducted according to VICH Good Clinical Practice (Wood et al., 1995) and WAAVP guidelines (Duncan et al., 2002), with approval granted by the University of New England’s Animal Ethics Committee. No adverse reactions to the BioWorma treatment were observed in any supplemented sheep throughout any of the trials.
An overview of the individual trials is presented in Table 1.
| Trial Number | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| Trial Dates | 27/1/2009 to 24/3/2009 | 22/11/2011–20/3/2012 | 16/10/2012 to 21/2/2013 | 14/12/2012 to 18/4/2013 |
| Location (Australia) | New England (Northern Tablelands) | Southern Tablelands | New England (Northern Tablelands) | South-east Queensland |
| Seasons | Summer/autumn | Spring/summer | Spring/summer | Summer/autumn |
| Pasture Type | Improved and native grass species and legumes
|
Improved and native grass species, predominantly phalaris, fescue, white clover and native poa tussock | Improved and native grass species, predominantly phalaris, fescue and white clover | Native grass species, such as Red Grass and Pitted Blue Grass |
| Seeder Sheep (breed, sex, age at start of grazing, number per group) | Merino, female, 6–18 months, 20 | Merino, male castrate, 12–16 months, 30 | Merino, male castrate and female, 6–24 months, 30 | Merino, male castrate and female, 6–24 months, 30 |
| Duration of grazing of seeder sheep (days) | 56 | 120 | 120 | 125 |
| Trigger threshold for salvage drench (epg) | N/A | 2000 | 1000 | 1000 |
| Tracer lambs (breed, sex, age at start of grazing, number per group) | Merino, male castrate, 4–6 months, 23 (10 lightest sacrificed for TWC) | Merino, male castrate and female, 6–12 months, 10 | Merino, male castrate and female, 6–12 months, 10 | Merino, male castrate and female, 6–12 months, 10 |
| Time of introduction of tracer lambs
|
End (following removal of seeder sheep) | Groups A, B: Midpoint
Groups C, D: End |
Groups A, B: Midpoint
Groups C, D: End |
Groups A, B: Midpoint
Groups C, D: End |
| Duration of tracers in pens prior to sacrifice (days) | 20 | Groups A, B: 12
Groups C, D: 11 |
Groups A, B: 12
Groups C, D: 12 |
Groups A, B: 12
Groups C, D: 13 |
Data were entered into Microsoft EXCEL (and verified following entry) and EXCEL used to calculate group summary data including arithmetic and geometric mean FECs, TWCs and bodyweights. Statistical analyses were performed using Statistix 8.0 and 9.0 (Analytical Software 2008 and 2010). Means were compared in the first instance using parametric Analysis of Variance (and Tukey’s All Pairwise Comparison Test) or 2-sample T-Tests; homogeneity of variances was not assumed. If variances appeared to be unevenly distributed (based on Levene’s Test) the equivalent non-parametric tests (primarily Kruskal–Wallis Analysis of Variance) were used. Means were compared in all cases at P < 0.05. The same statistical methodology was used in all 4 studies.
Total parasitic burden for each tracer animal was measured by TWCs while for seeder sheep the parasite burden of each animal was estimated from FECs. Speciation was based on larval differentiation and overall TWCs following sacrifice. Treatment efficacy was calculated according to the formula:
Efficacy = [Mean (Control) – Mean (Treated)]/[Mean (Control)] × 100 (1)
The supplements offered (BioWorma and placebo) were both well-accepted by the animals in each trial. Over all four trials, 99.4% of each of the BioWorma and Livamol supplements that were offered were consumed.
Group mean FECs of seeder sheep in Trials 1–4 are detailed in Table 2. FECs of seeder sheep in the Control and BioWorma groups in all trials were not significantly different (P < 0.05) at the time of allocation to groups (Week-1). In Trial 1, up to Week 6 there were no significant differences in FEC between groups, but by Week 8 the mean FEC for Group 1 (Control) was significantly higher than that for Group 2 (BioWorma). In Trial 2, the FEC for the BioWorma group was significantly lower (P < 0.05) from Week 11 onwards, despite the salvage drenches applied to the Control group. In Trial 3, significant differences were seen at Weeks 0 and 6, but not from Week 8 onwards and in Trial 4 significant differences were seen at Weeks 0, 6 and 11. It should be noted that in Trials 3 and 4 the data was impacted by the number of salvage drenches applied.
Table 2. Group mean FECs (epg) of seeder sheep in Trials 1–4 (Arithmetic means ± SE).
| Trial | Treatment | Week | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| -1 | 0 | 2 | 4 | 6 | 8 | 11 | 13 | 15 | END | ||
| 1 | Group 1 (Control)
|
598.0(1) ± 109.6 | - | 574.0(1)
± 107.1 |
1150.6(1)
± 218.0 |
322.2(1)
± 53.1 |
1292.0(1)
± 234.1 |
- | - | - | - |
| Group 2 (BioWorma) | 590.0(1)
± 111.8 |
- | 798.0(1)
± 112.0 |
1219.0(1)
± 198.0 |
292.6(1)
± 62.5 |
424.0(2)
± 115.6 |
- | - | - | - | |
| 2 | Group 1 (Control) | 905.2(1)
± 99.3 |
466.7(1) ± 44.5 | 192.3(1)
± 23.7 |
172.9(1)
± 29.3 |
288.3(1)
± 35.3 |
409.3(1)
± 86.3 |
608.3(1)
± 79.3 |
358.6(1)
± 61.3 |
366.7(1)
± 60.6 |
393.3(1)
± 47.4 |
| Group 2 (BioWorma) | 888.6(1)
± 102.8 |
413.3(1)
± 58.2 |
276.0(1)
± 45.5 |
313.1(2)
± 47.4 |
326.9(1)
± 40.4 |
408.6(1)
± 73.0 |
281.4(2)
± 37.3 |
208.0(2)
± 29.0 |
286.9(1)
± 40.1 |
261.3(2)
± 34.3 |
|
| 3 | Group 1 (Control) | 477.3(1)
± 86.1 |
649.7(1)
± 76.2 |
295.7(1)
± 47.7 |
408.0(1)
± 77.9 |
176.9(1)
± 32.2 |
224.6(1)
± 38.9 |
731.4(1)
± 112.9 |
620.8(1)
± 131.6 |
735.7(1)
± 215.3 |
682.7(1)
± 219.8 |
| Group 2 (BioWorma) | 445.3(1)
± 68.3 |
404.3(2)
± 69.3 |
416.0(1)
± 46.9 |
314.7(1)
± 43.5 |
296.6(2)
± 30.4 |
276.0(1)
± 36.4 |
678.5(1)
± 109.4 |
945.2(1)
± 146.0 |
590.0(1)
± 83.5 |
616.0(1)
± 120.0 |
|
| 4 | Group 1 (Control) | 236.0(1)
± 39.9 |
249.3(1)
± 32.8 |
338.7(1)
± 60.5 |
284.0(1)
± 34.7 |
789.3(1)
± 85.3 |
414.3(1)
± 86.7 |
326.7(1)
± 89.7 |
354.7(1)
± 117.8 |
209.3(1)
± 79.1 |
76.0(1)
± 33.3 |
| Group 2 (BioWorma) | 206.7(1)
± 28.5 |
458.7(2)
± 79.9 |
352.0(1)
± 39.2 |
238.7(1)
± 36.1 |
312.0(2)
± 49.3 |
278.7(1)
± 41.4 |
1194.5(2)
± 206.0 |
631.7(1)
± 167.0 |
247.1(1)
± 69.4 |
258.6(1)
± 91 |
|
(1), (2) Means within the same column and means type with the same numbered bracket are NOT significantly different at P < 0.05.
Group mean bodyweights are shown in Table 3. In Trial 1 there was a significantly higher weight gain in the BioWorma group by Week 8, which was consistent with their lower worm status as indicated by FECs. For Trials 2 and 3, no significant differences were seen between the treatment groups throughout the study. Bodyweights for Trial 4 sheep increased throughout the study for both groups with significant differences seen between treatment groups at Weeks 0 and 4. The Control group was significantly heavier at the commencement of the study as a consequence of the allocation to groups based on FEC. Since the animals were drawn from a single mob with a narrow age range, this may have introduced a slight unintended bias in favour of the Control group in terms of vigour. By the end of the study there was no significant difference between the groups.
The number of salvage treatments required in Trials 2, 3 and 4 are detailed in Table 4 (salvage treatments were not included in the design of Trial 1). In each trial the number of salvage treatments required by the BioWorma group was less than or equal to the Control group.
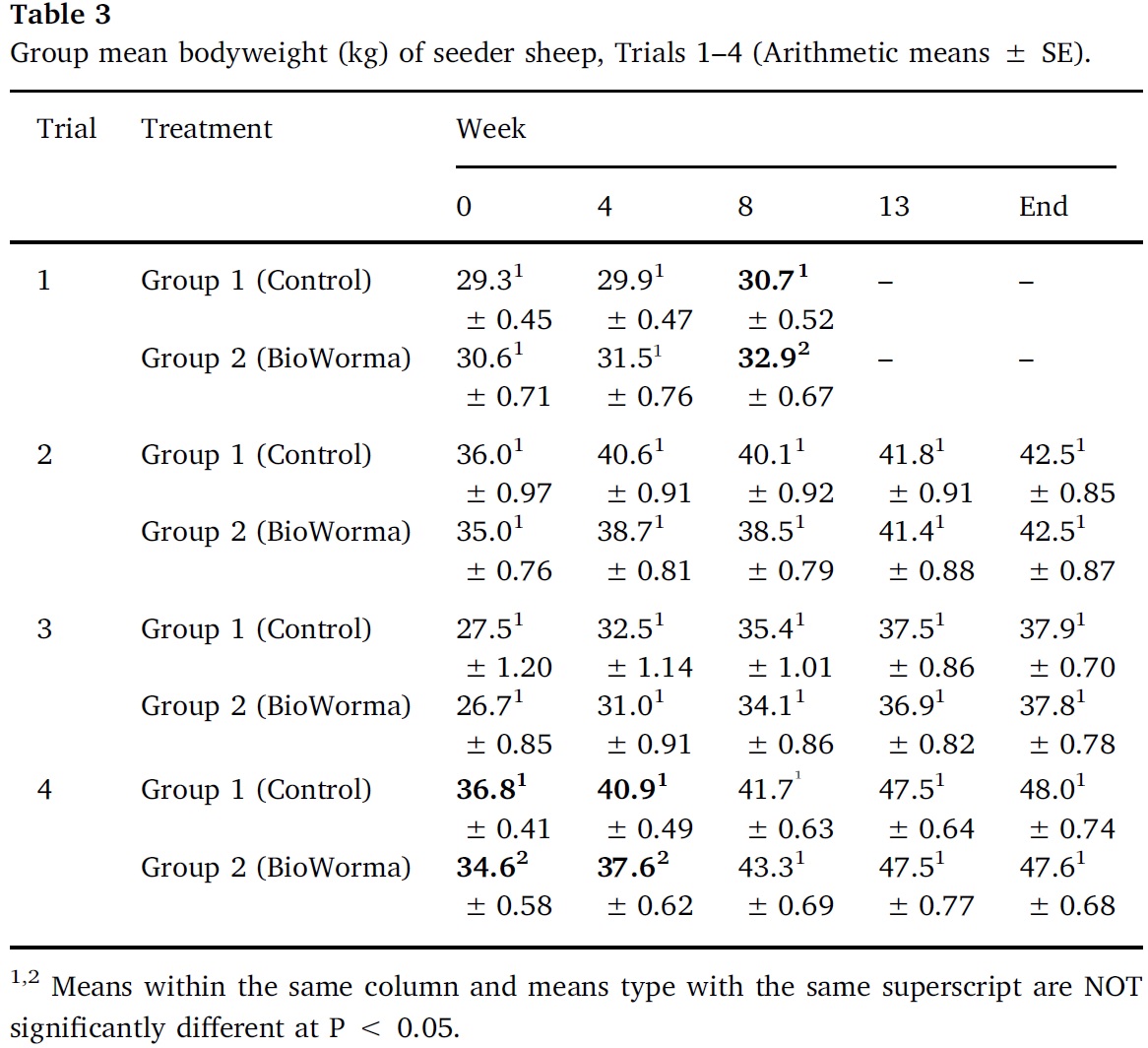
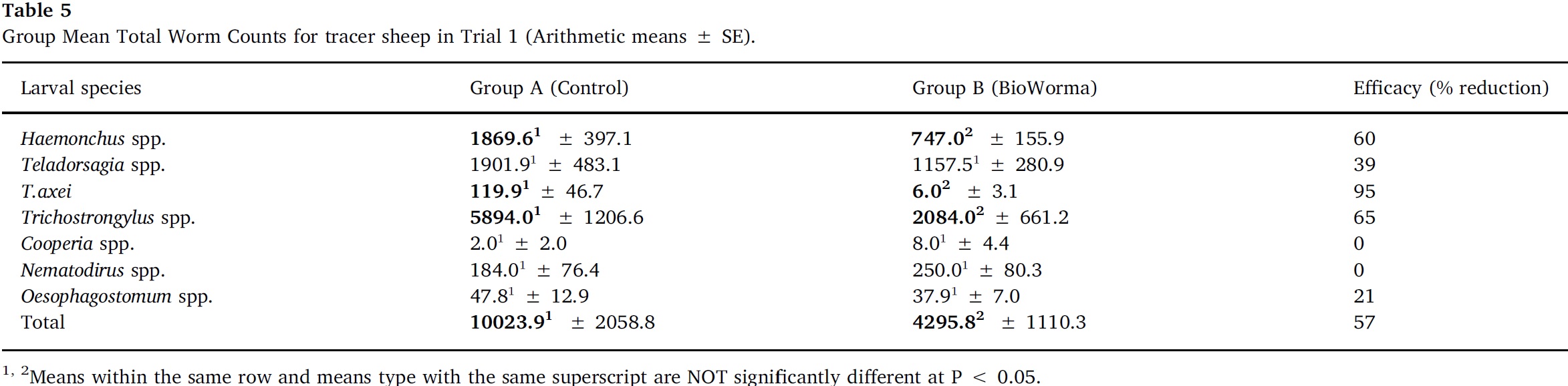
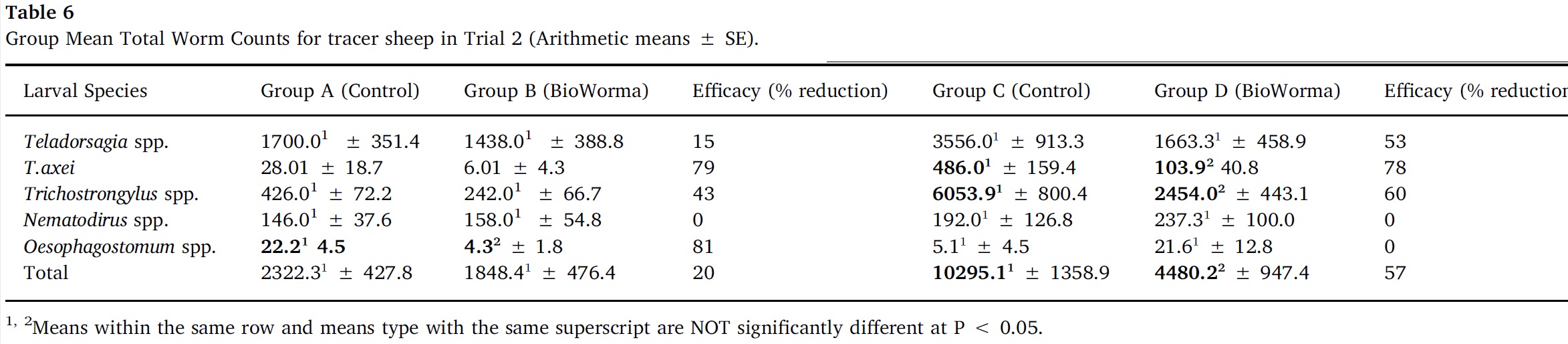
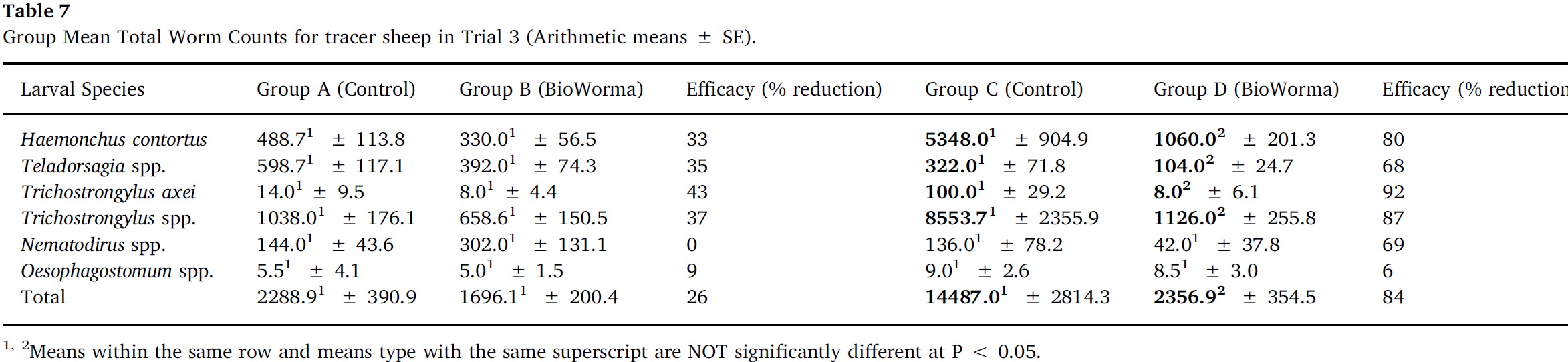
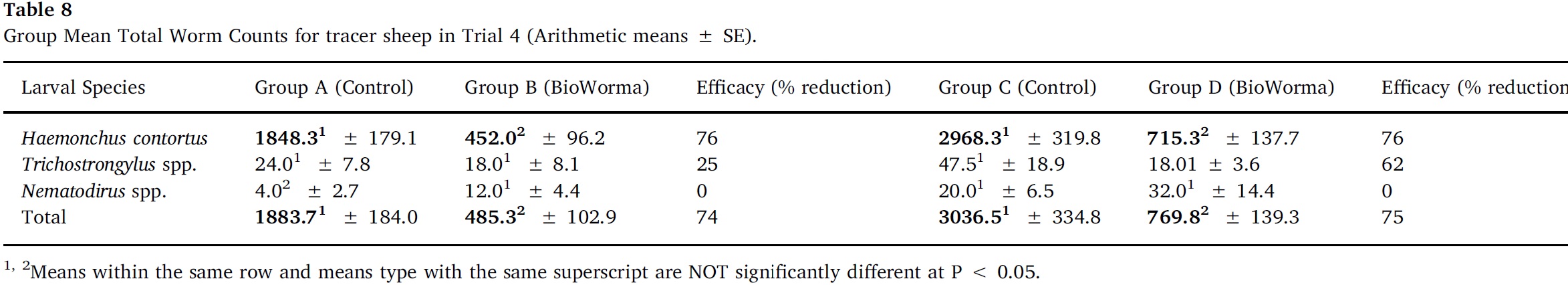
Arithmetic group means for overall TWC and for each worm species in Trials 1–4 are presented in Table 5, Table 6, Table 7, Table 8. In Trial 1, significant reductions due to BioWorma were seen in overall TWC by 57%, as well as Haemonchus spp. (60%), Trichostrongylus spp. (65%), and Trichostrongylus axei (95%). In Trial 2, significant reductions due to BioWorma were seen in overall TWC (57%), as well as Trichostrongylus spp. (60%) and Trichostrongylus axei (78%) in those tracers introduced to trial paddocks at the end-point of the study (i.e. Groups C/D). In Trial 3, significant reductions due to BioWorma were seen in overall TWC (84%) as well as Haemonchus spp. (80%), Trichostrongylus spp. (87%), Trichostrongylus axei (92%) and Teladorsagia spp. (68%) in those tracers introduced at the end-point of the study (i.e. Groups C/D). In Trial 4 significant reductions due to BioWorma were seen in overall TWC (74%) and Haemonchus spp. (76%) in those tracer sheep introduced at the mid-point of the study (Groups A/B) and in overall TWC (75%), and Haemonchus spp. (76%) in those tracer sheep introduced at the end point of the study (Groups C/D).
This series of four placebo-controlled studies carried out in Australia evaluated the effect of BioWorma (providing D. flagrans strain IAH 1297 at 3 × 104 chlamydospores/kg b.w./day) when administered to sheep harbouring naturally acquired worm burdens. Infections consisted predominantly of Haemonchus spp., Trichostrongylus spp. and Teladorsagia spp., including multi-resistant strains. The results demonstrated the ability of BioWorma to reduce the infectivity of pasture, as evidenced by 57–84% reduction in worm burdens in tracer sheep after grazing trial pastures. The results were consistent in sheep across a range of climatic zones and during different seasons.
In conclusion, these studies have shown that supplementation with BioWorma substantially reduced the infectivity of the pasture on which the recipient sheep grazed. Duration of treatment can affect efficacy as seen in Trials 2 and 3, where higher efficacy was observed at the end of the trials, by which time GIN numbers were higher in the tracer sheep. BioWorma provides a biological method to control worm burdens in grazing sheep by decreasing re-infection through pasture and will add another option for inclusion in integrated control programs to assist sheep producers to address this insidious problem. As this technology is most effectively used as a preventative means of GIN control in sheep, producers should plan BioWorma’s use while continuing to apply recommended good practice with regular FEC monitoring, and judicious use of anthelmintic chemicals when necessary.
These trials were funded by International Animal Health Products Pty Ltd, with additional support from the Australian Commonwealth Government (R&D Start, Commercial Ready and Early Stage Commercialisation grant schemes).
The authors acknowledge the contribution of Anne Tavares of International Animal Health Products Pty Ltd for proof reading.